Vascular Endothelial Growth Factor (VEGF) - signaling
VASCULAR ENDOTHELIAL GROWTH FACTORS (VEGFs) are the most important regulators of vessel morphogenesis. Not only do they participate in the regulation of both vasculogenesis and angiogenesis, but they also are among the most important molecules involved in the pathogenesis of angiogenic diseases, such as diabetic retinopathy and cancer. In endothelial cells, upon VEGF-A binding, phosphorylation of VEGFR2 on Tyr-1175 leads to recruitment of PLC-γ, which in turn, via activation of PKC, phosphorylates mitogen-activated protein kinase (MAPK)/ extracellular signal-regulated kinase1/2 (ERK1/2) leading to proliferation of ECs or leads to activation of the PI3K/AKT pathway, which is considered the main survival pathway of VEGF-A in ECs, as AKT conveys antiapoptotic signals. Phosphorylation of Tyr1214 of VEGFR2 activates stress-activated protein kinase-2/p38 leading to VEGF-A-induced actin reorganization and migration of ECs.
VEGF Signals through ATF6 and PERK to Promote Endothelial Cell Survival and Angiogenesis in the Absence of ER Stress
Accumulation of unfolded proteins in the endoplasmic reticulum (ER) initiates IRE1a, ATF6, and PERK cascades, leading to a transcriptional/translational response known as unfolded protein response (UPR). We have shown that VEGF activates UPR mediators through a PLCg-mediated crosstalk with the mTORC1 complex without accumulation of unfolded proteins in the ER. Activation of ATF6 and PERK contributes to the survival effect of VEGF on endothelial cells by positively regulating mTORC2-mediated phosphorylation of AKT on Ser473, which is required for full activity of AKT.
)
A schematic model of the identified VEGF signaling pathway that leads to activation of IRE1a, ATF6, and PERK via PLCg/mTORC1 pathway and enhancement of endothelial cell survival and angiogenesis.
E Karali et al., Molecular cell 54 (4), 559 (2014)

VEGF autoregulates its proliferative and migratory ERK1/2 and p38 cascades by enhancing the expression of DUSP1 and DUSP5 phosphatases in endothelial cells

We have demonstrated that VEGF strongly induces the transcription of two dual-specificity phosphatase (DUSP) genes DUSP1 and DUSP5 in endothelial cells. Gain-of-function and lack-of-function approaches revealed that DUSP1/MKP-1 dephosphorylates primarily VEGF-phosphorylated p38, thereby inhibiting endothelial cell migration, whereas DUSP5 dephosphorylates VEGF-phosphorylated ERK1/2 inhibiting proliferation of endothelial cells. Moreover, DUSP5 exhibited considerable nuclear anchoring activity on ERK1/2 in the nucleus, thereby diminishing ERK1/2 export to the cytoplasm decreasing its further availability for activation.
DUSP1/MKP-1 is localized in both the nucleus and cytoplasm of ECs, whereas DUSP5 is a strictly nuclear protein and dephosphorylates primarily VEGF-phosphorylated p38, thereby inhibiting EC migration,
S Bellou et al., Am J Physiol Cell Physiol ( ) (2009).
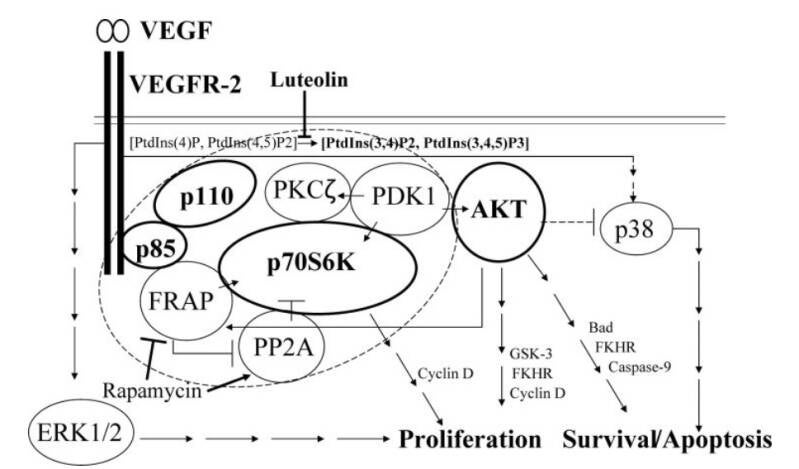
Mechanism of phytochemical action on VEGF signalling - Luteolin effect on VEGF signalling
We have shown that the flavonoid luteolin significantly inhibits tumor growth and angiogenesis by disrupting VEGF-induced survival and proliferation of endothelial cells in murine xenograft models and rabbit corneal assays. This is achieved by targeting the PI3K/Akt pathway, specifically blocking survival signals mediated by Akt and mitotic progression regulated by p70 S6 kinase. These findings highlight luteolin's potential as a phytochemical agent for suppressing VEGF-signalling essential for tumor development.
Flow diagram of the consequences of PI3K inhibition by luteolin on signaling cascades of VEGF regulating survival and proliferation in ECs.
E Bagli et al., Cancer research 64 (21), 7936 (2004)
Mechanism of phytochemical action on VEGF signalling / 6methoxyequol effect on VEGF signalling
We have screened isoflavonoids for anti-angiogenic activity and identified that 6-methoxyequol inhibits VEGF-induced MEK1/2 phosphorylation and endothelial cell proliferation leaving unaffected the migratory and survival functions of VEGF. Injecting directly 6-ME to the xenograft tumors, to bypass the low bioavailability, resulting in a statistically significant reduction of tumor volume compared to controls and suppressed vascularization.
Effect of 6-ME on VEGF-induced phosphorylation of MEK1/2 and ERK1/2
S Bellou et al., Molecular cancer 11, 35 (2012)
